주메뉴
- About IBS 연구원소개
-
Research Centers
연구단소개
- Research Outcomes
- Mathematics
- Physics
- Center for Underground Physics
- Center for Theoretical Physics of the Universe (Particle Theory and Cosmology Group)
- Center for Theoretical Physics of the Universe (Cosmology, Gravity and Astroparticle Physics Group)
- Dark Matter Axion Group
- Center for Artificial Low Dimensional Electronic Systems
- Center for Quantum Nanoscience
- Center for Exotic Nuclear Studies
- Center for Van der Waals Quantum Solids
- Center for Relativistic Laser Science
- Center for Trapped Ion Quantum Science
- Chemistry
- Life Sciences
- Center for Memory and Glioscience (Cognitive Glioscience Group)
- Center for Memory and Glioscience (Learning and Memory Group)
- Center for Synaptic Brain Dysfunctions
- Center for RNA Research
- Center for Genomic Integrity
- Center for Vascular Research
- Center for Genome Engineering
- Center for Microbiome–Body–Brain Physiology
- Earth Science
- Interdisciplinary
- Center for Neuroscience Imaging Research (Neuro Technology Group)
- Center for Neuroscience Imaging Research (Cognitive and Computational Neuroscience Group)
- Center for Algorithmic and Robotized Synthesis
- Center for Nanomedicine
- Center for Biomolecular and Cellular Structure
- Center for 2D Quantum Heterostructures
- Institutes
- Korea Virus Research Institute
- News Center 뉴스 센터
- Career 인재초빙
- Living in Korea IBS School-UST
- IBS School 윤리경영


주메뉴
- About IBS
-
Research Centers
- Research Outcomes
- Mathematics
- Physics
- Center for Underground Physics
- Center for Theoretical Physics of the Universe (Particle Theory and Cosmology Group)
- Center for Theoretical Physics of the Universe (Cosmology, Gravity and Astroparticle Physics Group)
- Dark Matter Axion Group
- Center for Artificial Low Dimensional Electronic Systems
- Center for Quantum Nanoscience
- Center for Exotic Nuclear Studies
- Center for Van der Waals Quantum Solids
- Center for Relativistic Laser Science
- Center for Trapped Ion Quantum Science
- Chemistry
- Life Sciences
- Center for Memory and Glioscience (Cognitive Glioscience Group)
- Center for Memory and Glioscience (Learning and Memory Group)
- Center for Synaptic Brain Dysfunctions
- Center for RNA Research
- Center for Genomic Integrity
- Center for Vascular Research
- Center for Genome Engineering
- Center for Microbiome–Body–Brain Physiology
- Earth Science
- Interdisciplinary
- Center for Neuroscience Imaging Research (Neuro Technology Group)
- Center for Neuroscience Imaging Research (Cognitive and Computational Neuroscience Group)
- Center for Algorithmic and Robotized Synthesis
- Center for Nanomedicine
- Center for Biomolecular and Cellular Structure
- Center for 2D Quantum Heterostructures
- Institutes
- Korea Virus Research Institute
- News Center
- Career
- Living in Korea
- IBS School
News Center
|
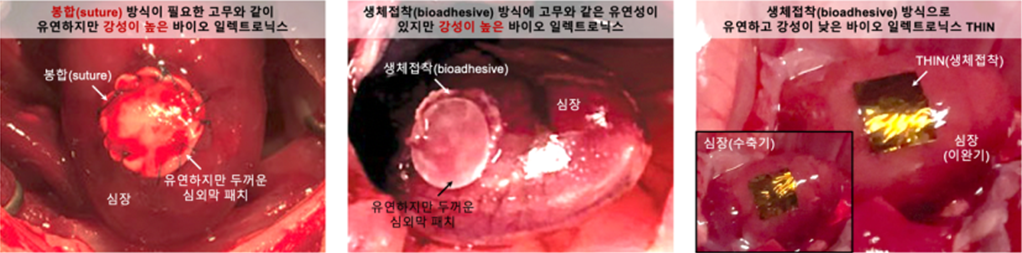
[Science Knowledge Encyclopedia] Transformer Bioelectronics THIN, 1/300 the Thickness of a Human HairApplication of Bio Electronics in Living Tissues Have you ever imagined an electronic device so thin and soft that you would not even feel it attached to your body? Bioelectronics technology, which can be attached to organs or the skin to read signals, diagnose diseases, and even perform treatment, is becoming an important component of future medicine. Recently, a research team at the Institute for Basic Science (IBS) Center for Neuroscience Imaging Research reported new possibilities by developing “transformer bioelectronics THIN,” which is only 1/300 the thickness of a human hair, yet becomes rigid when dry and transforms into a gel when exposed to water. In this article, the participating researchers explain the significance of this study and the key concepts needed to understand it. What is wearable healthcare?Wearable healthcare refers to technologies worn on or attached to the body to measure and manage health in real time. Devices such as smartwatches that monitor heart rate, patch-type glucose sensors, and skin-attached temperature patches continuously track biological signals in daily life and help manage health. With advances in information and communication technology (ICT) and growing interest in personalized medicine, wearable healthcare has become a rapidly growing field. Because these technologies must operate reliably during movement and daily activities, flexible electronic materials and devices that can bend, stretch, and deform are essential. As wearable healthcare advances, there is an increasing need for more precise and reliable biological information that cannot be obtained from the skin surface alone. To monitor high-resolution signals—such as cardiac electrical patterns, fine brain signals, and neural transmission in muscles—over long periods in daily life, new technologies are needed in which electronic devices adhere more closely to biological tissue or are directly attached to organ surfaces. However, because the human body is soft and constantly moving, conventional rigid electronic devices have limitations in adapting to these conditions. Research addressing these challenges is driving next-generation soft bioelectronics, which is considered important for maintaining competitiveness in advanced bio technologies. How can bioelectronics be applied to biological tissues?Recently, experiments by Neuralink, which involve inserting electrodes into the brain to read and interpret neural signals, have attracted significant attention. Technologies like this are representative examples of bioelectronics that directly interface with biological tissue to measure or control neural activity, opening new possibilities in medical, rehabilitation, and assistive engineering fields. Technologies based on direct interfaces with biological tissue are attracting attention because they can extend high-resolution biological signal measurement—previously limited to hospital settings—into everyday life. To achieve this, a key challenge is how stably electronic devices can adhere to biological tissue and maintain their function over time. Because human tissue is soft, wet, irregular, and constantly moving, conventional rigid silicon-based electronics tend to detach or slip easily, leading to reduced signal quality. To address this, new approaches have been proposed. One such approach replaces rigid materials with soft, stretchable rubber-like materials, known as “intrinsically stretchable” electronics. While this approach improves mechanical flexibility, it does not fully solve the challenge of long-term stable attachment to tissue surfaces. Conventional methods often rely on sutures, staples, or physical fixation to attach devices to the body, which can cause tissue damage and lead to inflammation or biocompatibility issues over time. To overcome these limitations, bioelectronics based on “bioadhesion,” which can attach directly to tissue, have emerged as a potential solution. This approach has been actively studied, with representative examples including bioelectronics using intrinsically stretchable bioadhesive semiconductor materials reported in Science in 2023. However, previously reported bioadhesive intrinsically stretchable bioelectronics still require additional substrates to form devices, meaning the material cannot function independently. As a result, overall device thickness increases, and despite low material stiffness, effective bending stiffness becomes high. This highlights the need for continued research and development in both materials and fabrication processes.
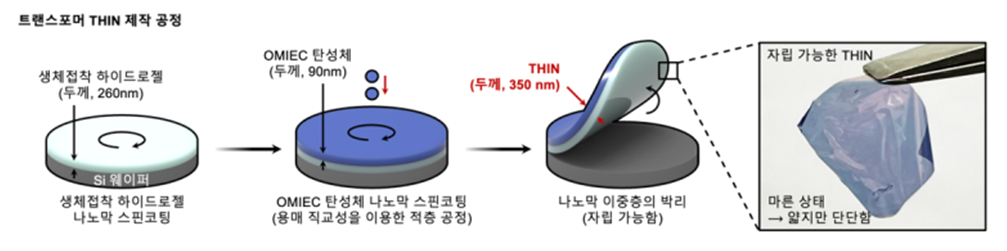
Development of THIN, transformer bioelectronics at 1/300 the thickness of a hairPaper: Hydrogel–elastomer-based conductive nanomembranes for soft bioelectronics, Nature Nanotechnology (2025) To reduce the effective bending stiffness of bioadhesive intrinsically stretchable bioelectronics, the IBS Center for Neuroscience Imaging Research team focused on developing materials and device processes that maintain sufficient mechanical stability for handling even at extreme thinness while preserving high transconductance. The team created an ultrathin amphiphilic nanomembrane with a total thickness of 350 nm by integrating a bioadhesive layer (260 nm) and an ionic–electronic semiconductor layer (organic mixed ionic–electronic conductor, OMIEC; 90 nm) into a single structure. Based on this, they developed a biosignal amplification sensor using an organic electrochemical transistor (OECT) with a new structural concept called THIN (transformable and imperceptible hydrogel–elastomer adhesive bilayer based on ionic–electronic conductive nanomembranes). Unlike previous bioelectronics, THIN can be handled without a separate substrate, allowing the distance between the semiconductor channel and biological tissue to be reduced to ~400 nm when hydrated. This maximizes the efficiency (transconduction) of signal transfer from tissue to the device.
The newly developed THIN-based OECT (THIN-OECT) overcomes structural and mechanical limitations of existing bioelectronics. When hydrated, its bending stiffness decreases by approximately 100 million times (9.08×10⁻⁵ GPa·μm⁴), and its mechanical stability (hardness) transitions from 1.35 GPa to a soft state close to biological tissue (35 MPa), showing distinctive transformation properties. Because of its ultrathin structure, the strain energy per unit area when hydrated is extremely low (0.0032 mN·m⁻¹), allowing it to conform naturally to fine curvatures (~1 μm) of biological tissue. In addition, the bioadhesive layer based on catechol chemistry (mussel-inspired chemistry) improves the work of adhesion at the tissue interface, enabling stable physical and chemical adhesion even on curved surfaces.
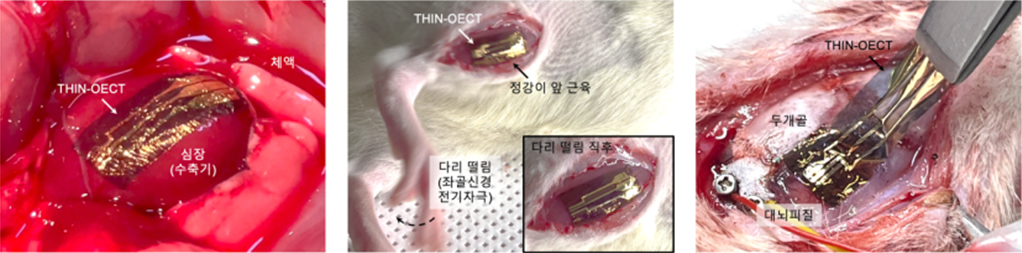
In terms of electrical performance, THIN-OECT operates at a low voltage (−0.6 V) while maintaining high transconductance (338 S·cm⁻¹). The µC* value (µ: charge mobility; C*: volumetric capacitance), a key metric for OMIEC materials, reached 1,034 F·cm⁻¹·V⁻¹·s⁻¹, significantly improving performance compared to previously developed materials. The material also shows excellent mechanical stability, with no degradation in electrical properties or signal amplification even when stretched up to 200%. Using THIN-OECT-based biosignal amplification sensors, the researchers successfully measured high-quality electrophysiological signals—including epicardial electrogram (EGM), electromyogram (EMG), and electrocorticogram (ECoG)—from various biological tissues, and confirmed long-term biocompatibility. These results were reported in Nature Nanotechnology (impact factor 35.1).
Application of THIN to next-generation bioelectronics?The THIN-based bioelectronics developed by the research team maintain high ionic–electronic transduction performance despite being only 1/300 the thickness of a human hair, and show excellent mechanical adaptability by conforming to fine curvature and movement of biological tissue when hydrated. In particular, the ability to self-adhere to tissue when hydrated, to be handled without a substrate, and to achieve high-performance signal amplification at low voltage represents a meaningful advance over existing biosensors. Beyond being simply a “thin sensor,” this technology represents a new type of biointerface platform that minimizes the boundary between biological tissue and electronic devices, and may help accelerate the development of personalized biohealthcare. The research team continues follow-up studies to further improve the technology and aims to contribute to maintaining competitiveness in advanced bio technologies in the near future. |
| before |
|---|
- Content Manager
- Public Relations Team : Suh, William Insang 042-878-8137
- Last Update 2023-11-28 14:20










![[Science Knowledge Encyclopedia] Transformer Bioelectronics THIN, 1/300 the Thickness of a Human Hair Application of Bio Electronics in Living Tissues](https://www.ibs.re.kr/dext5data/2026/03/20260305_103829061_45257.png)