주메뉴
- About IBS 연구원소개
-
Research Centers
연구단소개
- Research Outcomes
- Mathematics
- Physics
- Center for Underground Physics
- Center for Theoretical Physics of the Universe (Particle Theory and Cosmology Group)
- Center for Theoretical Physics of the Universe (Cosmology, Gravity and Astroparticle Physics Group)
- Dark Matter Axion Group
- Center for Artificial Low Dimensional Electronic Systems
- Center for Quantum Nanoscience
- Center for Exotic Nuclear Studies
- Center for Van der Waals Quantum Solids
- Center for Relativistic Laser Science
- Center for Trapped Ion Quantum Science
- Chemistry
- Life Sciences
- Center for Memory and Glioscience (Cognitive Glioscience Group)
- Center for Memory and Glioscience (Learning and Memory Group)
- Center for Synaptic Brain Dysfunctions
- Center for RNA Research
- Center for Genomic Integrity
- Center for Vascular Research
- Center for Genome Engineering
- Center for Microbiome–Body–Brain Physiology
- Earth Science
- Interdisciplinary
- Center for Neuroscience Imaging Research (Neuro Technology Group)
- Center for Neuroscience Imaging Research (Cognitive and Computational Neuroscience Group)
- Center for Algorithmic and Robotized Synthesis
- Center for Nanomedicine
- Center for Biomolecular and Cellular Structure
- Center for 2D Quantum Heterostructures
- Institutes
- Korea Virus Research Institute
- News Center 뉴스 센터
- Career 인재초빙
- Living in Korea IBS School-UST
- IBS School 윤리경영


주메뉴
- About IBS
-
Research Centers
- Research Outcomes
- Mathematics
- Physics
- Center for Underground Physics
- Center for Theoretical Physics of the Universe (Particle Theory and Cosmology Group)
- Center for Theoretical Physics of the Universe (Cosmology, Gravity and Astroparticle Physics Group)
- Dark Matter Axion Group
- Center for Artificial Low Dimensional Electronic Systems
- Center for Quantum Nanoscience
- Center for Exotic Nuclear Studies
- Center for Van der Waals Quantum Solids
- Center for Relativistic Laser Science
- Center for Trapped Ion Quantum Science
- Chemistry
- Life Sciences
- Center for Memory and Glioscience (Cognitive Glioscience Group)
- Center for Memory and Glioscience (Learning and Memory Group)
- Center for Synaptic Brain Dysfunctions
- Center for RNA Research
- Center for Genomic Integrity
- Center for Vascular Research
- Center for Genome Engineering
- Center for Microbiome–Body–Brain Physiology
- Earth Science
- Interdisciplinary
- Center for Neuroscience Imaging Research (Neuro Technology Group)
- Center for Neuroscience Imaging Research (Cognitive and Computational Neuroscience Group)
- Center for Algorithmic and Robotized Synthesis
- Center for Nanomedicine
- Center for Biomolecular and Cellular Structure
- Center for 2D Quantum Heterostructures
- Institutes
- Korea Virus Research Institute
- News Center
- Career
- Living in Korea
- IBS School
News Center
Scientists Engineer a Tool to “Edit” Brain Circuits and Enhance Memory- A new molecular tool called SynTrogo harnesses astrocytes to selectively dismantle synaptic connections - Every thought, memory, and feeling we experience depends on trillions of tiny connection points in the brain called synapses. These are the junctions where one neuron passes signals to another, forming the vast communication network known as the connectome—the brain’s wiring diagram. Although scientists have developed powerful tools to increase or decrease neural activity, directly redesigning the brain’s physical wiring has remained far more difficult. A research team led by Dr. LEE Sangkyu and Director C. Justin LEE at the Center for Memory and Glioscience within the Institute for Basic Science (IBS), in collaboration with Dr. LEE Kea Joo of the Korea Brain Research Institute (KBRI), has now developed a molecular tool that makes such structural editing possible. The new platform, called SynTrogo (Synthetic Trogocytosis), enables researchers to induce astrocytes to selectively remodel synaptic connections in a targeted brain circuit. The brain already has a natural mechanism for refining its wiring. During development and throughout life, unneeded or weak connections are removed in a process known as synaptic pruning, much like trimming unnecessary branches from a tree. This pruning is partly carried out by astrocytes—star-shaped glial cells that closely surround synapses and help maintain the neural environment. When this process becomes dysregulated, either through too much or too little pruning, it has been linked to disorders such as schizophrenia, autism spectrum disorder, and Alzheimer’s disease. Until now, however, there has been no method for deliberately triggering this kind of structural remodeling at a chosen location in the brain without also altering the electrical activity of the circuit. Existing techniques such as optogenetics and chemogenetics can modulate how strongly neurons fire, but they largely act on synapses that are already there. By contrast, SynTrogo is designed to act on the physical connections themselves. The system works like a molecular lock-and-key mechanism. Neurons in the target circuit are engineered to display a molecular “tag” on their surface (a lock), while nearby astrocytes are engineered with a matching binding partner (a key). When the two cells come into contact, the astrocyte is induced to “nibble” part of the neuronal membrane and nearby synaptic material through a trogocytosis-like process—a form of partial cellular uptake seen in several biological systems. By harnessing this process synthetically, the researchers created a way to selectively reduce synaptic connectivity in a defined neural circuit. Dr. LEE Sangkyu, Junior Chief Investigator at the IBS Center for Memory and Glioscience said, “This is the first demonstration that brain circuits can be directly edited by engineering physical interactions between neurons and astrocytes, independent of neuronal activity. It opens the possibility of ‘connectome editing’ and provides a new platform for studying and reshaping the physical architecture of neural circuits.” To test the method in the brain, the team applied SynTrogo to the hippocampus, a region essential for learning and memory. They expressed the synthetic ligand in CA3 excitatory neurons and the matching receptor in CA1 astrocytes, targeting one of the best-known memory circuits in the mammalian brain. Three weeks later, the number of excitatory synapses in the targeted region had decreased by about 27 percent. At first glance, fewer synapses might be expected to impair brain function. However, the researchers found the opposite. The surviving synapses became structurally and functionally stronger: presynaptic boutons enlarged, synaptic vesicle numbers increased, and the remaining connections transmitted signals more efficiently. Electrophysiological experiments further showed that long-term potentiation (LTP)—a key cellular process underlying learning and memory—was significantly enhanced. The remaining synapses also exhibited coordinated remodeling on both the presynaptic and postsynaptic sides, and rather than causing widespread degeneration, the process appeared to be spatially restricted and self-limiting. To directly examine the structural basis of these changes, the researchers used high-resolution imaging techniques, including correlative light and electron microscopy. These analyses revealed unusually tight interfaces between astrocytes and neuronal axons, along with localized membrane deformation and partial enclosure of axonal regions. Consistent with the functional results, targeted axons contained fewer synapses, while the remaining synapses displayed clear ultrastructural remodeling. This finding was especially important because it showed that reducing synapse number does not necessarily mean weakening the circuit. Instead, the brain appears capable of compensatory refinement: when some connections are removed, the remaining ones can reorganize and become more effective. Dr. LEE Kea Joo, Principal Investigator at KBRI said, “We found that the brain can adapt and strengthen its function even when the total number of synaptic connections is reduced. This gives us new insight into how neural circuits maintain performance and may offer clues for restoring cognitive function in brain disorders.” The team then asked whether these cellular changes translated into behavioral effects. In contextual fear-conditioning experiments, mice with SynTrogo-modified hippocampal circuits showed stronger memory than control animals. They displayed enhanced recall both two days after learning and 23 days later, indicating improvements in both recent and remote memory. Importantly, these mice also remained capable of extinction learning—the process by which previously learned fear responses are reduced when they are no longer appropriate—suggesting that SynTrogo strengthened memory without sacrificing cognitive flexibility. Further analysis suggested that SynTrogo may place synapses into a more plastic, learning-ready state. Before learning, AMPA receptor-mediated synaptic responses were reduced, but after fear conditioning they recovered to control-like levels. This implies that the remodeled circuit may be particularly poised for experience-dependent strengthening when new learning occurs. Director C. Justin LEE of the IBS Center for Memory and Glioscience said, “This study shows that selectively reducing a subset of synapses can paradoxically enhance circuit function by promoting adaptive remodeling of the remaining connections. It also lays the groundwork for a new strategy to study and potentially treat brain disorders associated with abnormal synapse numbers.” Beyond its value as a neuroscience tool, SynTrogo may have broad biomedical implications. Many neurological and psychiatric disorders—including autism spectrum disorder, schizophrenia, Alzheimer’s disease, and brain injury—have been associated with abnormal synapse numbers or disrupted circuit organization. Yet researchers have lacked a way to directly manipulate synaptic connectivity in living mammalian brains with cell-type specificity. By enabling targeted structural remodeling through engineered neuron-astrocyte interactions, SynTrogo provides a foundation for future efforts in connectome editing and may eventually help guide therapeutic strategies aimed at restoring healthy circuit function. Perhaps the most fundamental message of the study is that the brain is not simply strongest when it has the greatest possible number of synapses. Under some conditions, removing selected connections can trigger the network to reorganize itself in a more efficient and adaptable way. This resilience has long been difficult to isolate experimentally. SynTrogo now offers a way to study that process directly. The study was published online in Nature Communications in 2026. 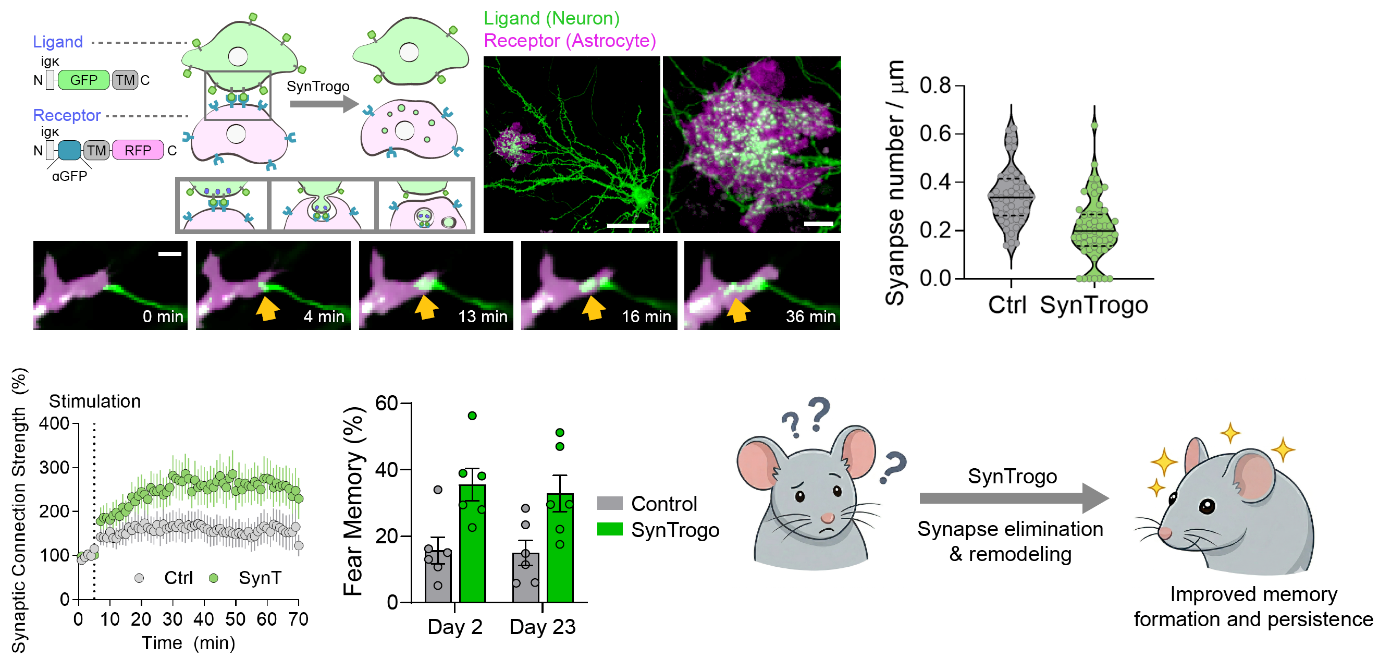 Figure 1. Synthetic Trogocytosis (SynTrogo) enables the "nibbling" of neuronal membranes by astrocytes through engineered ligand and receptor proteins. Upon SynTrogo induction, the synaptic density of the target CA3-CA1 hippocampal circuit was significantly reduced by ~27%. Conversely, the remaining synapses underwent structural and functional remodeling – characterized by the enlargement of pre- and post-synaptic compartments, enhanced long-term potentiation (LTP), and improved memory formation and retention. Notes for editors
- References
- Media Contact
- About the Institute for Basic Science (IBS) |
| before |
|---|
- Content Manager
- Public Relations Team : Suh, William Insang 042-878-8137
- Last Update 2023-11-28 14:20











