주메뉴
- About IBS 연구원소개
-
Research Centers
연구단소개
- Research Outcomes
- Mathematics
- Physics
- Center for Underground Physics
- Center for Theoretical Physics of the Universe (Particle Theory and Cosmology Group)
- Center for Theoretical Physics of the Universe (Cosmology, Gravity and Astroparticle Physics Group)
- Dark Matter Axion Group
- Center for Artificial Low Dimensional Electronic Systems
- Center for Quantum Nanoscience
- Center for Exotic Nuclear Studies
- Center for Van der Waals Quantum Solids
- Center for Relativistic Laser Science
- Center for Trapped Ion Quantum Science
- Chemistry
- Life Sciences
- Center for Memory and Glioscience (Cognitive Glioscience Group)
- Center for Memory and Glioscience (Learning and Memory Group)
- Center for Synaptic Brain Dysfunctions
- Center for RNA Research
- Center for Genomic Integrity
- Center for Vascular Research
- Center for Genome Engineering
- Center for Microbiome–Body–Brain Physiology
- Earth Science
- Interdisciplinary
- Center for Neuroscience Imaging Research (Neuro Technology Group)
- Center for Neuroscience Imaging Research (Cognitive and Computational Neuroscience Group)
- Center for Algorithmic and Robotized Synthesis
- Center for Nanomedicine
- Center for Biomolecular and Cellular Structure
- Center for 2D Quantum Heterostructures
- Institutes
- Korea Virus Research Institute
- News Center 뉴스 센터
- Career 인재초빙
- Living in Korea IBS School-UST
- IBS School 윤리경영


주메뉴
- About IBS
-
Research Centers
- Research Outcomes
- Mathematics
- Physics
- Center for Underground Physics
- Center for Theoretical Physics of the Universe (Particle Theory and Cosmology Group)
- Center for Theoretical Physics of the Universe (Cosmology, Gravity and Astroparticle Physics Group)
- Dark Matter Axion Group
- Center for Artificial Low Dimensional Electronic Systems
- Center for Quantum Nanoscience
- Center for Exotic Nuclear Studies
- Center for Van der Waals Quantum Solids
- Center for Relativistic Laser Science
- Center for Trapped Ion Quantum Science
- Chemistry
- Life Sciences
- Center for Memory and Glioscience (Cognitive Glioscience Group)
- Center for Memory and Glioscience (Learning and Memory Group)
- Center for Synaptic Brain Dysfunctions
- Center for RNA Research
- Center for Genomic Integrity
- Center for Vascular Research
- Center for Genome Engineering
- Center for Microbiome–Body–Brain Physiology
- Earth Science
- Interdisciplinary
- Center for Neuroscience Imaging Research (Neuro Technology Group)
- Center for Neuroscience Imaging Research (Cognitive and Computational Neuroscience Group)
- Center for Algorithmic and Robotized Synthesis
- Center for Nanomedicine
- Center for Biomolecular and Cellular Structure
- Center for 2D Quantum Heterostructures
- Institutes
- Korea Virus Research Institute
- News Center
- Career
- Living in Korea
- IBS School
Research Centers
- Research Outcomes
- Mathematics
- Physics
- Center for Underground Physics
- Center for Theoretical Physics of the Universe (Particle Theory and Cosmology Group)
- Center for Theoretical Physics of the Universe (Cosmology, Gravity and Astroparticle Physics Group)
- Dark Matter Axion Group
- Center for Artificial Low Dimensional Electronic Systems
- Center for Quantum Nanoscience
- Center for Exotic Nuclear Studies
- Center for Van der Waals Quantum Solids
- Center for Relativistic Laser Science
- Center for Trapped Ion Quantum Science
- Chemistry
- Life Sciences
- Center for Memory and Glioscience (Cognitive Glioscience Group)
- Center for Memory and Glioscience (Learning and Memory Group)
- Center for Synaptic Brain Dysfunctions
- Center for RNA Research
- Center for Genomic Integrity
- Center for Vascular Research
- Center for Genome Engineering
- Center for Microbiome–Body–Brain Physiology
- Earth Science
- Interdisciplinary
- Center for Neuroscience Imaging Research (Neuro Technology Group)
- Center for Neuroscience Imaging Research (Cognitive and Computational Neuroscience Group)
- Center for Algorithmic and Robotized Synthesis
- Center for Nanomedicine
- Center for Biomolecular and Cellular Structure
- Center for 2D Quantum Heterostructures
- Institutes
- Korea Virus Research Institute
Illuminating the brain functions by investigating the stars in the brain, astrocyte
Tel. +82-42-878-8234
Tel. +82-42-878-9155 (lab)
IBS Center for Memory and Glioscience, Room C211, 55 Expo-ro, Doryong-dong, Yuseong-gu, Daejeon (34126), South Korea
Major Publications

- - Astrocytes Control Sensory Acuity via Tonic Inhibition in the Thalamus
- - Severe reactive astrocytes precipitate pathological hallmarks of Alzheimer's disease via H2O2-production
- - Aberrant Tonic Inhibition of Dopaminergic Neuronal Activity Causes Motor Symptoms in Animal Models of Parkinson's Disease
- - Non-invasive optical control of endogenous Ca2+ channels in awake mice
- - Neural circuits underlying a psychotherapeutic regimen for fear disorders
Director

Director LEE C. Justin
Dr. LEE is the co-director of the Center for Memory and Glioscience, established in July 2012. He earned his B.A. in Chemistry from The University of Chicago before getting his Ph.D. from Columbia University in 2001, and later worked in Department of Pharmacology at Emory University as a postdoctoral fellow. In 2004 he joined KIST as a senior research scientist and later served as the Director of Center for Neuroscience. In 2009, he founded the WCI Center for Functional Connectomics as a part of World Class Institute Program. In 2015 he became the recipient of Creative Research Investigator Award to establish the Center for Glia-Neuron Interaction at KIST to serve as the Director of the Center before taking up his position in IBS.
Introduction

Investigating the brain function in regulating cognition and sociality
The Center for Memory and Glioscience (CCS) focuses on unraveling the mechanisms of brain function for cognition and sociality, identifying causes of psychiatric disorders and neurodegenerative diseases, and developing novel treatments through research at various levels encompassing molecules, cells, and organisms. The CCS consists of eight laboratories in three groups (Cognitive Glioscience, Social Neuroscience, and Molecular Neuroimaging Groups) according to the research topics. We are conducting multidisciplinary research based on genetics, behavioral genetics, electrophysiology, optogenetics, acoustic neuromodulation, molecular neuro-imaging, brain wave analysis, synthetic biology, and glycomics.
1. Cognitive Glioscience Group
The Cognitive Glioscience Group focuses on research to understand and regulate brain cognitive functions. We have been investigating the role of astrocytes in cognitive functions and seeking to find answers to questions about how various gliotransmitters such as Glutamate, D-serine, GABA, BDNF, and H2O2 are synthesized and secreted from astrocytes to regulate brain cognitive function, how astrocytic volume changes can regulate synaptic plasticity, and how reactive astrocytes can cause neurodegenerative diseases such as Alzheimer's disease, Parkinson’s disease and Huntington’s disease. Through these studies, we aim to clarify the importance of astrocytes in brain cognitive function and discover drug candidates that can regulate the function of astrocytes, thereby presenting a new paradigm for the treatment of various neurodegenerative diseases and neuropsychiatric disorders.
For translational research, we are developing ultrasonic technologies for non-invasive neuromodulation. We are particularly interested in the fundamental molecular and cellular mechanism of how ultrasound is sensed and transduced by various ion channels and transporters. We believe that this approach will provide an unprecedented way for selective and specific control of brain functions to treat neurological and psychiatric diseases.
In addition, we are trying to develop translational and reverse-translational research behavioral paradigm to understand how human brain as well as animal brain encode the abstract information by processing complex sensory signals. We deploy multiple techniques such as ECoG, EEG, PET/CT, and MRI to understand how various perceptual, cognitive, and conscious processes unfold in time.
2. Social Neuroscience Group
The Social Neuroscience Group is studying the brain mechanisms that control a variety of social behaviors and emotions. We are particularly interested in the neural mechanisms of “empathy,” the ability to share and understand the emotional state of others. Using an observational fear model that assesses affective empathy in rodents, we seek to find mechanistic explanation for how the brain generates the affect sharing, and how pathological dysfunction within these brain networks causes abnormal empathic responses. Understanding neural mechanisms of observational fear will provide novel insights into effective treatments for psychiatric disorders associated with empathy.
Our group is also conducting research on neural underpinnings of how the brain recognizes individuals as unique identities during social interactions. We have been developing simplified and precisely controlled novel individual discrimination paradigms. Together with quantitative behavioral measures, we use multiple state-of-the-art techniques including two-photon calcium imaging, miniscope imaging, and Neuropixels recordings to reveal neural mechanisms of social recognition.
In addition, we are pioneering new fields of research to uncover the role of glycosylation of proteins and lipids in brain function for social behaviors. We have been generating a variety of genetically engineered mouse models targeting glycan-modifying enzymes and building a brain map of glycan structures. These efforts will broaden our understanding of the molecular mechanisms of glycosylation to regulate social behaviors and contribute to the development of new diagnostic methods and treatments for various neuropsychiatric disorders such as autism, depression, and schizophrenia.
3. Molecular Neuroimaging Group
The Molecular Neuroimaging Group has been developing molecular technologies for real-time visualization or control of brain functions at the molecular level. We are developing genetically encoded fluorescent biosensors to monitor dynamic molecular interactions and the activity of specific proteins in the brain cells from freely moving animals. Also, we are designing a variety of optogenetic tools for precise modulation of specific molecules in space and time by light illumination. With the aid of various fluorescence imaging instruments such as confocal microscopes, two-photo microscopes, super-resolution microscopes, light-sheet microscopes, FACS, we are studying brain functions on various scales from nanometers (10-9 m) to centimeters (10-2 m). Currently, our group mainly focuses on the development of optogenetic technologies for control of various channel proteins in the brain and new synthetic approaches for modulating the brain connections to decipher the meaning of communication between brain cells and clarify the structure-function relationship of brain circuits.
Organization
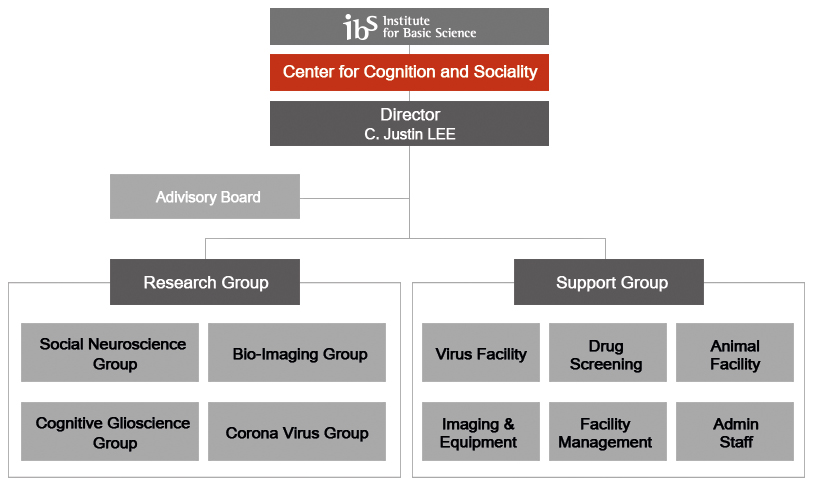
Main research results
- Astrocytes Control Sensory Acuity via Tonic Inhibition in the Thalamus
(Neuron, 2020) - Severe reactive astrocytes precipitate pathological hallmarks of Alzheimer's disease via H2O2-production
(Nature Neuroscience, 2020) - Aberrant Tonic Inhibition of Dopaminergic Neuronal Activity Causes Motor Symptoms in Animal Models of Parkinson's Disease
(Current Biology, 2020) - Non-invasive optical control of endogenous Ca2+ channels in awake mice
(Nature Communications, 2020) - Neural circuits underlying a psychotherapeutic regimen for fear disorders
(Nature, 2019)
Personnel
| Total | 78 |
|---|---|
| Gender | 48(Male), 30(Female) |
| Korean/ International | 72(Korean), 6(International) |
Degree
Position
As of April. 2021
- Content Manager
- Operational Support Team : Park Jung Won 042-878-8403
- Last Update 2026-03-20 17:46











