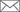
▲ In vitro transcribed-CRISPR guide RNAs trigger innate immune response in cells, but can be prevented by removing the triphosphate moiety. Credit: Sojung Kim
February 22, 2018 - CRISPR-mediated genome editing has become a powerful tool for modeling of disease in various organisms and is being developed for clinical applications. Preassembled Cas9 ribonucleoproteins (RNPs) composed of the recombinant Cas9 protein and in vitro transcribed (IVT) guide RNA complexes can be delivered into cells without risk of foreign DNA integration into the host genome and with fewer off-target effects. However, in a study published today in Genome Research, scientists discovered in vitro-transcribed gRNAs, containing a 5' triphosphate (5'ppp) moiety, activate the immune response in human cells leading to cell death.
"CRISPR-Cas9 RNPs are now broadly used in biomedical research and hold promise in therapeutic genome editing" says corresponding author Jin-Soo Kim from the Center for Genome Engineering within the Institute for Basic Science (IBS) (Seoul, South Korea). Thus, translation of this technology from biomedical applications to clinical applications requires a thorough investigation into potentially deleterious host responses.
In this study, researchers generated and transfected IVT gRNAs complexed with purified Cas9, into human cervical cancer cells (HeLa) and mouse embryonic fibroblasts. In these cells, 5'ppp gRNAs elicited a type I interferon-mediated immune response, as well as increased expression of interferon-activated antiviral effector proteins, such as DDX58, resulting in cell death.
Additionally, this study demonstrates the cytotoxic effects of 5'ppp gRNA on human T cells. "Genome editing in T cells holds great promise in cell therapy for the treatment of cancer and HIV infection. T cells were extremely sensitive to the 5' triphosphate group in gRNAs" says Kim. The T cell surface receptor, CCR5, is involved in HIV virus entry and is therefore a promising target for anti-HIV therapies. Kim et al. demonstrated that pretreatment of IVT gRNAs with phosphatase, removes the 5'ppp and significantly reduces T cell host immune response and cytotoxicity. Phosphatase treated IVT gRNAs displayed increased mutation efficiency against CCR5 resulting in significantly decreased cell surface presentation of this HIV coreceptor.
This research elucidates the cytotoxic effects on human cells by immune activating IVT gRNAs in CRISPR-Cas9 or Cpf1 ribonucleoprotein complexes and offers a new method to prevent host immune response, while maintaining high mutation efficiency, toward the effective therapeutic application of CRISPR-mediated genome editing.
Researchers from the IBS Center for Genome Engineering, University of Science and Technology, National Medical Center, and Seoul National University contributed to this work. The study was funded by a grant from the Institute for Basic Science (IBS-R021-D1).
The press release is written by Jennifer L. DeLeon, Ph.D. Assistant Editor, Genome Research
Notes for editors
- References
The manuscript will be published online ahead of print on 22 Februray 2018. Its full citation is as follows:
Kim S, Koo T, Jee H, Cho H, Lee G, Lim D, Shik Shin H, and Kim J. 2018. CRISPR RNAs trigger innate immune responses in human cells. Genome Res doi: 10.1101/gr.231936.117
- Media Contacts:
The authors are available for more information by contacting Dahee Carol Kim, Institute for Basic Science Communications team (clitie620@ibs.re.kr); +82-42-878-8133 [office]).
Interested reporters may obtain copies of the manuscript via email from Dana Macciola, Administrative Assistant, Genome Research (macciol@cshl.edu); +1-516-422-4012).
- About the Institute for Basic Science (IBS)
IBS was founded in 2011 by the government of the Republic of Korea with the sole purpose of driving forward the development of basic science in South Korea. IBS has launched 28 research centers as of February 2018. There are nine physics, one mathematics, six chemistry, eight life science, one earth science, and three interdisciplinary research centers.