주메뉴
- About IBS 연구원소개
-
Research Centers
연구단소개
- Research Outcomes
- Mathematics
- Physics
- Center for Theoretical Physics of the Universe(Particle Theory and Cosmology Group)
- Center for Theoretical Physics of the Universe(Cosmology, Gravity and Astroparticle Physics Group)
- Center for Exotic Nuclear Studies
- Center for Artificial Low Dimensional Electronic Systems
- Center for Underground Physics
- Center for Axion and Precision Physics Research
- Center for Theoretical Physics of Complex Systems
- Center for Quantum Nanoscience
- Center for Van der Waals Quantum Solids
- Chemistry
- Life Sciences
- Earth Science
- Interdisciplinary
- Institutes
- Korea Virus Research Institute
- News Center 뉴스 센터
- Career 인재초빙
- Living in Korea IBS School-UST
- IBS School 윤리경영


주메뉴
- About IBS
-
Research Centers
- Research Outcomes
- Mathematics
- Physics
- Center for Theoretical Physics of the Universe(Particle Theory and Cosmology Group)
- Center for Theoretical Physics of the Universe(Cosmology, Gravity and Astroparticle Physics Group)
- Center for Exotic Nuclear Studies
- Center for Artificial Low Dimensional Electronic Systems
- Center for Underground Physics
- Center for Axion and Precision Physics Research
- Center for Theoretical Physics of Complex Systems
- Center for Quantum Nanoscience
- Center for Van der Waals Quantum Solids
- Chemistry
- Life Sciences
- Earth Science
- Interdisciplinary
- Institutes
- Korea Virus Research Institute
- News Center
- Career
- Living in Korea
- IBS School
News Center
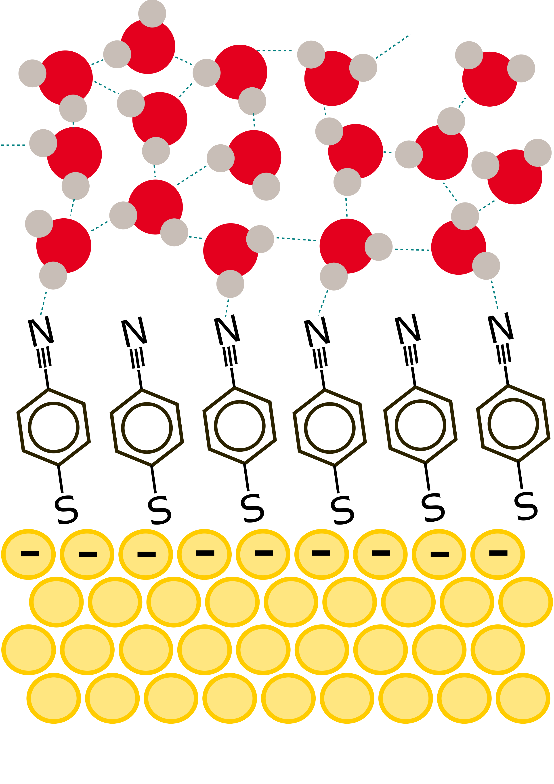
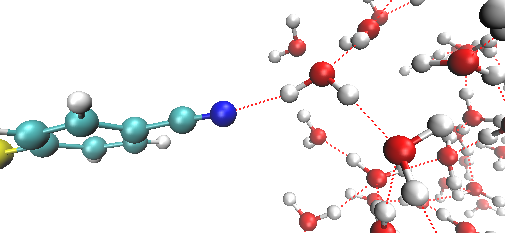
First observation of how water molecules move near a metal electrode- Movements of water molecules change depending on the applied voltage - A collaborative team of experimental and computational physical chemists from South Korea and the United States have made an important discovery in the field of electrochemistry, shedding light on the movement of water molecules near metal electrodes. This research holds profound implications for the advancement of next-generation batteries utilizing aqueous electrolytes. In the nanoscale realm, chemists typically utilize laser light to illuminate molecules and measure spectroscopic properties to visualize molecules. However, studying the behavior of water molecules near metal electrodes proved challenging due to the overwhelming interference from metal atoms in the electrode itself. Additionally, water molecules distant from the electrode surface also contribute to the response of the applied light, complicating the selective observation of molecules at the liquid-metal electrode interface. Led by Professor Martin Zanni from the University of Wisconsin at Madison and Director CHO Minhaeng from the Center for Molecular Spectroscopy and Dynamics within the Institute for Basic Science (IBS) addressed this challenge with newly developed spectroscopic techniques coupled with computer imulations. In order to minimize the interference from the metals, the authors coated the surface of the electrode with specially designed organic molecules. Then, surface-enhanced femtosecond (10-15 second) twodimensional vibrational spectroscopy was employed to observe the changes in the movement of water molecules near the metal electrode. Depending on the magnitude and polarity of the applied voltage on the metal electrode, the researchers observed, for the first time, either a deceleration or acceleration of the motion of water molecules near the electrode. “When a positive voltage is applied to the electrode, the movement of nearby water molecules slows down. Conversely, when a negative voltage is applied, the opposite is observed both in femtosecond vibrational spectroscopy and in computer simulations,” explains Dr. Kwac. “The results of this study provide crucial information for understanding electrochemical reactions, offering essential physical insights necessary for the research and development of aqueous electrolyte batteries in the future,” comments Director CHO Minhaeng of the IBS Center for Molecular Spectroscopy and Dynamics, a corresponding author of the study. This outcome implies a close relationship between electrochemical reactions involving water on the surface of electrodes and the dynamics of interfacial water molecules. It is expected to not only advance our understanding of fundamental electrochemical processes but also pave the way for the design of more efficient and sustainable battery technologies. This research was published in the Proceedings of the National Academy of Sciences (PNAS) on December 18, 2023.
Notes for editors
- References
- Media Contact
- About the Institute for Basic Science (IBS)
|
| Next | |
|---|---|
| before |
- Content Manager
- Public Relations Team : Suh, William Insang 042-878-8137
- Last Update 2023-11-28 14:20