주메뉴
- About IBS 연구원소개
-
Research Centers
연구단소개
- Research Outcomes
- Mathematics
- Physics
- Center for Underground Physics
- Center for Theoretical Physics of the Universe (Particle Theory and Cosmology Group)
- Center for Theoretical Physics of the Universe (Cosmology, Gravity and Astroparticle Physics Group)
- Dark Matter Axion Group
- Center for Artificial Low Dimensional Electronic Systems
- Center for Theoretical Physics of Complex Systems
- Center for Quantum Nanoscience
- Center for Exotic Nuclear Studies
- Center for Van der Waals Quantum Solids
- Center for Relativistic Laser Science
- Chemistry
- Life Sciences
- Earth Science
- Interdisciplinary
- Center for Neuroscience Imaging Research (Neuro Technology Group)
- Center for Neuroscience Imaging Research (Cognitive and Computational Neuroscience Group)
- Center for Algorithmic and Robotized Synthesis
- Center for Genome Engineering
- Center for Nanomedicine
- Center for Biomolecular and Cellular Structure
- Center for 2D Quantum Heterostructures
- Center for Quantum Conversion Research
- Institutes
- Korea Virus Research Institute
- News Center 뉴스 센터
- Career 인재초빙
- Living in Korea IBS School-UST
- IBS School 윤리경영


주메뉴
- About IBS
-
Research Centers
- Research Outcomes
- Mathematics
- Physics
- Center for Underground Physics
- Center for Theoretical Physics of the Universe (Particle Theory and Cosmology Group)
- Center for Theoretical Physics of the Universe (Cosmology, Gravity and Astroparticle Physics Group)
- Dark Matter Axion Group
- Center for Artificial Low Dimensional Electronic Systems
- Center for Theoretical Physics of Complex Systems
- Center for Quantum Nanoscience
- Center for Exotic Nuclear Studies
- Center for Van der Waals Quantum Solids
- Center for Relativistic Laser Science
- Chemistry
- Life Sciences
- Earth Science
- Interdisciplinary
- Center for Neuroscience Imaging Research (Neuro Technology Group)
- Center for Neuroscience Imaging Research (Cognitive and Computational Neuroscience Group)
- Center for Algorithmic and Robotized Synthesis
- Center for Genome Engineering
- Center for Nanomedicine
- Center for Biomolecular and Cellular Structure
- Center for 2D Quantum Heterostructures
- Center for Quantum Conversion Research
- Institutes
- Korea Virus Research Institute
- News Center
- Career
- Living in Korea
- IBS School
News Center
|
IBS Reports a High Performance Nanoparticle Electrocatalyst Scientists from the Center for Nanoparticle Research develop a high performance and cost effective fuel cell December 10, 2015
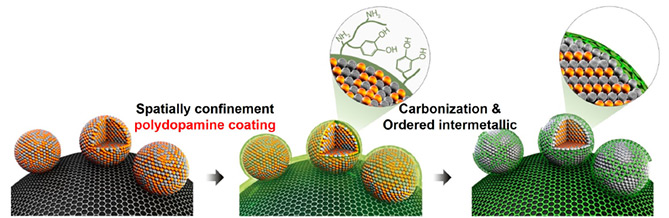
Scientists operating out of IBS’ Center for Nanoparticle Research have reported highly durable and active intermetallic platinum-iron (PtFe) nanoparticles (NPs) coated with nitrogen (N) doped carbon shell. Precision sized face centered tetragonal (fct) PtFe NPs, only a few nanometers thick, are formed by thermal annealing at 700oC, resulting in a carbon outer layer which protects the NPs from detachment and dissolution throughout the harsh fuel cell operating conditions. The N-doped carbon shell not only prevents the amalgamation of the NPs during a thermal annealing process to keep their sizes as small as 6.5 nm but also protects them under the harsh operating condition. Nanoparticles are microscopic particles between 1 and 100 nanometers (nm) in size. To put that into perspective, the greatest particle size that can pass through a surgical mask is 100 nm. In 1959, physicist Richard Feynman proposed that one day humankind could create machines composed of several individually manipulated molecules or atoms, and these machines could be constructed by tools that were only slightly larger -an inventive and, for the time, perplexing thought.
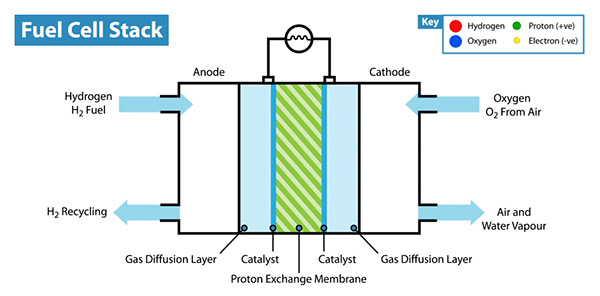
Rejuvenated Nano Research Now, some 40 years later, nanoparticle research has seen something of a renaissance and is an area of intense scientific research, due to a wide variety of potential applications in biomedical, optical, and electronic fields. Published reports have increased exponentially since 2000 and there’s little evidence to assume this trend will cease. Demand for a practical synthetic approach to the high performance electrocatalyst is rapidly increasing for fuel cell commercialization. An electrocatalyst is an electrical current that acts as a catalyst. A fuel cell is a device that converts chemical energy from a fuel into electricity through an electrochemical reaction of positively charged hydrogen ions with oxygen or another oxidizing agent - an oxidizing agent or an oxidizer is a chemical species that transfers electronegative atoms, usually oxygen, to a substrate. These devices are incredibly popular due to their ability to generate electricity by reacting oxygen and hydrogen without emitting CO2. Fuel cells are commonly found in all types of equipment and vehicles, the most common types are found in cars, airplanes, boats and military equipment like submarines and weaponry. There are however limitations to fuel cells: they require platinum which is expensive and found in limited deposits on Earth. Fuel Cells in Ordinary Life Nanoparticle-based electrocatalysts have been intensively investigated for fuel cell applications over the past decade, mainly motivated by their high mass activity. Great effort has been exhausted to utilize the high activity and surface area of NPs in order to make a breakthrough for fuel cell commercialization. The team’s paper, published in the Journal of the American Chemical Society, stated that a practical use of nanomaterials for fuel cell electrocatalyst is impeded by their low physical and chemical stability. Under the standard fuel cell operating conditions, NPs are often oxidized, dissolved, or detached from the support and clustered into larger particles, losing their electrochemical catalytic activity during cycling. Therefore, ordered intermetallic NPs are considered as one of the most promising candidates to achieve both high activity and stability in practical fuel cell applications. The resulting ordered tetragonal-PtFe/ C nanocatalyst coated with an N-doped carbon shell shows the higher performance and durability compared to disordered face centered cubic (fcc)-PtFe/C and commercial Pt/C. According to the team’s paper, their approach ‘can open a new possibility for the development of high performance and cost effective fuel cell catalysts. The paper entitled ‘Highly Durable and Active PtFe Nanocatalyst for Electrochemical Oxygen Reduction Reaction’ is published in the Journal of the American Chemical Society. By Neil Mannix Notes for editors - References - Media Contact - About the Institute for Basic Science (IBS)
|
|||
|
|
| Next | |
|---|---|
| before |
- Content Manager
- Public Relations Team : Yim Ji Yeob 042-878-8173
- Last Update 2023-11-28 14:20